Lesson Plans
Return to Lesson Plan Index
Printer Friendly Version
Condiment Diver: The World's Simplest Cartesian Diver
Grades: 6-8
Author: Diane Zak
Source: Condiment Diver: www.wildfiles.tv, Eric Muller: originally published in the Physics Teacher, May 1996
Abstract
This activity uses a condiment packet to teach students how fish use their swim bladders to rise and descend in the water. The students will also learn about density, buoyancy, and sinking and floating.
Objectives
What should students know as a result of this lesson?
- Students should be able to explain why some objects sink and some objects float.
- Students should be able to explain what density is and how it affects objects and other substances.
- Students should be able to explain why the condiment packet floats and what causes it to sink, and then relate this information to how fish, and submarines rise and descend in the water.
What should the students be able to do as a result of this lesson?
- Students should be able to form a hypothesis
- Students should be able to collect and analyze data
- Students should be able to communicate data by using a data collection sheet
- Students should be able to draw a conclusion based on the data collected
Materials
- Several squeeze condiment packets (soy sauce, ketchup, mustard, etc.) Many fast food restaurants, such as McDonald's, Arby's, and Burger King, will donate the condiments. * Not all condiments behave the same; therefore it is recommended that they be tested beforehand. Have twice the number of condiment packets that you think you will need.
- Clear plastic bottle with tight-fitting lid (1-liter is easier to use for small hands than 2-liter); Wide-mouth bottles are easier to use than regular water bottles in case it is necessary to remove the packet from the bottle.
- *Optional: a pair of tweezers to help retrieve the condiment packets from the bottle of water - when the condiment packet needs to be exchanged.
- Bucket or large container to empty the bottles of water - when the condiment packet needs to be exchanged.
- Bowl of water for testing packets. The teacher may want to put "catch pans" or trays under students' bottles in case of splashes or spills.
- Water to fill the plastic bottles: Gallon milk jugs work well
- Paper towels
- Worksheet
Procedures
Engagement
- Place a fresh egg in a clear glass of water. It should sink to the bottom. Ask the students how they can get the egg to float without touching the egg. (Gradually add salt. As more salt is added, the egg will begin to float. You can get the egg to remain in the center of the glass before it floats to the top. Sink and float at the same time - neutral buoyancy. By continuing to add salt, the density of the water is increasing. The egg will eventually float on top of the water.)
- The density of a fresh egg is greater than that of an older egg because it loses water through the tiny pores in the shell. (So, don't eat an egg that floats in fresh water. It's spoiled.) If you save an egg for several days past the expiration date, you will be able to demonstrate this to the students. Keep checking the egg at home to make sure it is ready to float in time for your demonstration. Place a fresh egg in water - it goes to the bottom and lies on its side; 3-4 days old - stays on the bottom at a 45 degree angle; 10-12 days old - stays on the bottom, but stands straight up; If it floats to the top - it is bad and should not be opened in the house because it will smell.
- Show a picture of a submarine or fish and ask the students what they know about its motion, what makes it possible for each to rise and sink in the water.


The swim bladder is filled with gasses which are produced in its blood. When the swim bladder is inflated with these gasses, it increases the fishes' volume decreasing its density, which keeps it from sinking.

Pictures from Holt Science & Technology (2005) Harcourt Education Company: Forces, Motion, and Energy: page 76 & 77
Assessment: Before proceeding, by your questions and their responses, make sure the students' understand what density is and how it affects objects.
- Explain how a fish is able to rise and sink in water
- Explain how a submarine is able to dive and float in water
- Explain how to check if an egg, past its expiration date, is still safe to eat
Exploration
Before beginning this activity, review safety procedures with students; make sure that students know they are not to taste/eat anything in the science lab.
1. Today we will make a condiment diver. Find out if your condiment packet is a good candidate for a Cartesian diver by dropping the packet in a bowl of water. (See "Content Knowledge" for an explanation of what a Cartesian diver is and how it is made.) The best ones just barely float. *Even though the packets just barely float, some of them still will not sink in the bottle when the bottle is squeezed.

2. Fill the clear plastic bottle with water leaving a small amount of air space. Push the unopened packet into the bottle.

Screw the cap on tight.
Squeeze the bottle to make the packet sink, and release to make it rise. *If the diver does not sink, use another packet. This is where a wide mouth opening is to your advantage since it makes it easier to remove the packet from the bottle. A pair of tweezers also works to help remove the packet from the bottle, but be careful not to puncture the packet.
Assessment: Monitor the students' work to make sure they are following the correct procedures, making observations, and recording data accurately.
Redirect their attention to the task, as needed. Make sure that students are employing safe practices as they conduct the experiment.
Check to see that each member of the group is participating.
Answer students' questions regarding procedures.
- Name the brands and the contents of the condiment packets that floated.
- Name the brands and the contents of the condiment packets that did not float.
- Name the brands and the contents of the condiment packets that worked as Cartesian divers.
- Are there any similarities with the condiment packets that worked as Cartesian divers.
- Is there anything inside the packets besides the condiment? (Air)
Explanation
Students report their findings.
- What property are we talking about when we are talking about objects floating? (Density which is a comparison between the mass of an object and its volume. Mass is how much stuff is in an object. Volume is how much space an object occupies. Density = mass/volume; Also Buoyant force: Water and air pressure increase the deeper you go. Think of a "Cheer-leader pyramid" - Who has more pressure on them, the person at the top or the cheerleaders on the bottom? (bottom since they have the rest of the cheerleaders on top of them) Since there is more pressure at the bottom of an object in water than at the top, there is an upward force on the object. This force is buoyant force.
If an object in water weighs more than the buoyant force, it will sink. If an object in water weighs the same as the buoyant force, it will float. The steel that is used to build a ship is much heavier and denser than water. So why does it float? The secret is its shape. It is built with a hollow shape which increases it's volume (how much space it takes up), which in turn reduces its density. Therefore it floats.)
- Why do you think the condiment packet floats? (The density of the packet is less than the density of the water.)
- What happens to the pressure on the inside of the bottle when you squeeze the bottle? (It increases)
- When the bottle is squeezed what changes, to the condiment packet, are taking place? (The air in the packet is compressed which changes the volume, which in turn changes the density.)
- What is affecting the buoyancy of the condiment packet? (Density of the packet/ air inside the packet.)
* Most students are familiar with air being in flotation devices for swimming. Relate this same principle to the condiment packets, which also contain some air.
Assessment: Listen to students' accounts of their findings to judge if their reports are supported by the findings that you observed as experiments were being conducted.
Ascertain students' knowledge of density and buoyancy by asking questions. See above # 1-5
- Have students create a concept map for buoyant force and density. Include information that shows objects that float on the surface of water, objects that float between the surface and the bottom, and objects that sink to the bottom.
- Name three things that can be done to change the density of an object. (Change its shape, change its mass or change its volume.)
Elaboration
- Take a 6 square inch piece of foil. Press it into the shape of a boat. Place it gently in a container of water. Record your observations. (It floats)
- Roll the foil boat into a ball. Place the foil ball gently into the container of water. Record your observations. (It sinks)
- What did you change that made the piece of foil sink? (Changed the shape, which decreased the volume, which increased the density, causing it to sink)
Literature:
- Swim Like a Fish by Ellen Schecter (1998) Gareth Stevens Publishing Milwaukee, Wisconsin
- Who Sank the Boat? By Pamela Allen (book about a cow, donkey, sheep, pig, and a mouse who decide to go for a boat ride.) (1988-1996) Putnam Publishing
- Early Step into Reading Boats by Shana Corey (2001) Random House New York
- I Love Boats by Flora McDonnell (1995) Candlewick Press Cambridge, Massachusetts
Assessment: Ask questions to ascertain students' understanding of density and buoyancy. Give students 2-3 minutes to complete a brief written summary of the experience. The prompt will be: Imagine that your friend tells you that all heavy objects sink in water. Explain why you agree or disagree with his statement.
Make a poster that explains how a life jacket helps a person float. (Most are made of porous material filled with air. The life jacket increases the person's volume which decreases his/her density, therefore the person floats.)
Prerequisites
No prerequisites required.
Best Teaching Practices
- Inquiry
- Hands-on Teaching
- Questioning
Alignment with Standards
NGSS Standards:
- MS-PS2-2 Plan an investigation to provide evidence that the change in an object's motion depends on the sum of the forces on the object and the mass of the object.
Common Core Standards:
- RST.6-8.3 Follow preciesly a multistep procedure when carrying our experiments, taking measurements, or performing technical tasks.
- WHST.6-8.2 Write informative/explanatory texts, including the narration of historical events, scientific procedures/experiments, or technical processes.
- WHST.6-8.9 Draw evidence from informational texts to support analysis, reflection, and research.
National Standards:
- Science as Inquiry Content Standard A, B, and G
- Physical Science Content Standard B
Ohio Standards:
- Scientific Inquiry
- Physical Science
Content Knowledge
- See Learning Cycle Procedure: The picture of the fishes' swim bladder and the ballast tanks in a submarine that enables it to rise and sink.
- The original Cartesian Diver is made with a pipette and a hex nut that can fit snugly onto the stem of the pipette. Plastic Cartesian Diver Pipettes from Educational Innovation , Inc. www.teachersource.com (2006) #pp-222A Bag of 100 $6.95 & Brass hex nuts #CD-3 Bag of 100 $9.95
1. Cut off almost all of the stem (leave about 15mm). Place the hex nut snugly onto the remaining part of the stem.
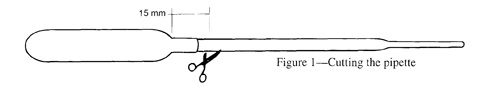
2. Fill the pipette about 1/2 -way with water. Place the pipette with the nut into a bottle of water. Screw the lid on tightly, squeeze the bottle.
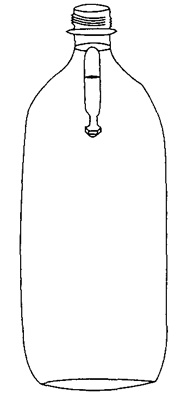
The pipette sinks to the bottom of the bottle when it is squeezed, and rises when you release the squeeze. If it doesn't work, adjust the amount of water in the pipette, by adding or subtracting water in the pipette.
- Whether an object sinks or floats is dependant on whether the object's density is greater or less than the liquid it is in.
- Since density is a comparison between the mass of the object to the volume of the object, if you add to the mass without changing its volume, the density will increase. If you keep the mass the same, but decrease the volume, the density will decrease. Since gas/air can be compressed, when you apply pressure to the bottle, you also apply pressure to the air in the packet which in turn will decrease the volume of the packet, making the packet less dense.
Density = mass /volume. A pan balance is the instrument used to find the mass of an object.
- Most of the sauces in the condiment packets are denser than water which should make the packets sink, but there is also some air in the packets.
- It is the air that helps the packets float. When you squeeze the bottle, you put pressure on the bottle, which increases the pressure on the water, which in turn increases pressure on the packet causing the air to compress. This compression makes the packet smaller, which causes the packet to sink/dive. When you release the pressure on the bottle, the packet returns to the original volume, causing it to rise.
- Submarines use the same principle to float and dive.
- Scuba divers add weights/mass to keep from floating.
- If the density of an object is less than the density of water, it will float. If the density of an object is greater than the density of water, it will sink.

Safety
Remind students to use materials for the intended purpose; no horseplay in the lab; do not eat or taste anything in the science lab. Be careful about spills; report any spills to the teacher.
Applications
Fish use swim bladders to rise and descend in the water.
Submarines use the same principle to rise and descend in the water.
Scuba divers use the principle of buoyancy and density to float and sink in the water.
Safety devices for swimming and boating such as life preservers, life raft, "air wings" for the upper arms of young children that cannot swim, etc.
Assessment
See Learning Cycle, Exploration, Elaboration Assessment, Worksheet
Other Considerations
Grouping Suggestions: Have each student make his/her own Condiment diver, but work in groups of two for the activities
Pacing/Suggested Time: Four class periods - depending on the age of the students and how much detail/involvement you require.
Printable PDF Worksheets
Condiment Worksheet








